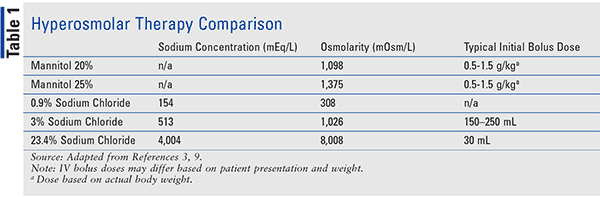
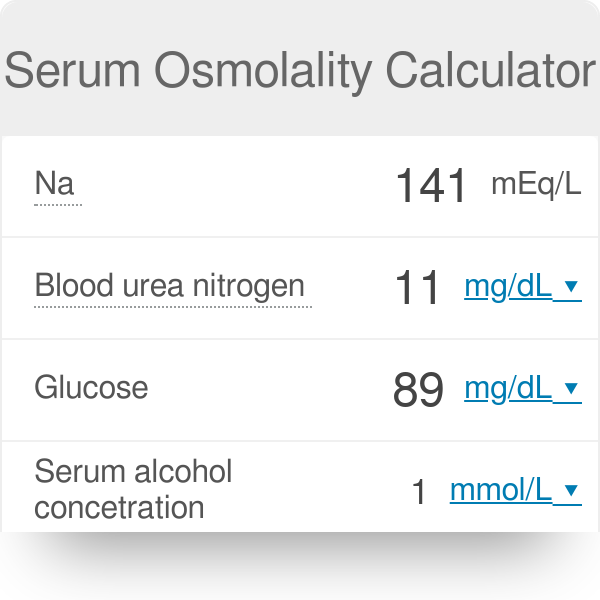
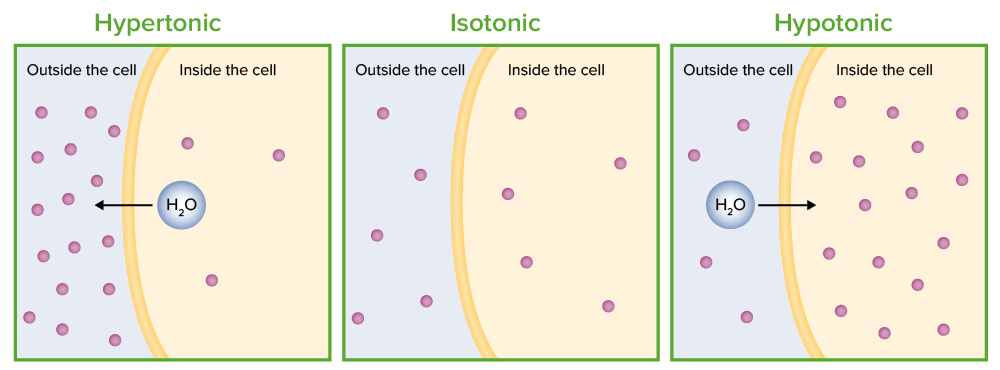
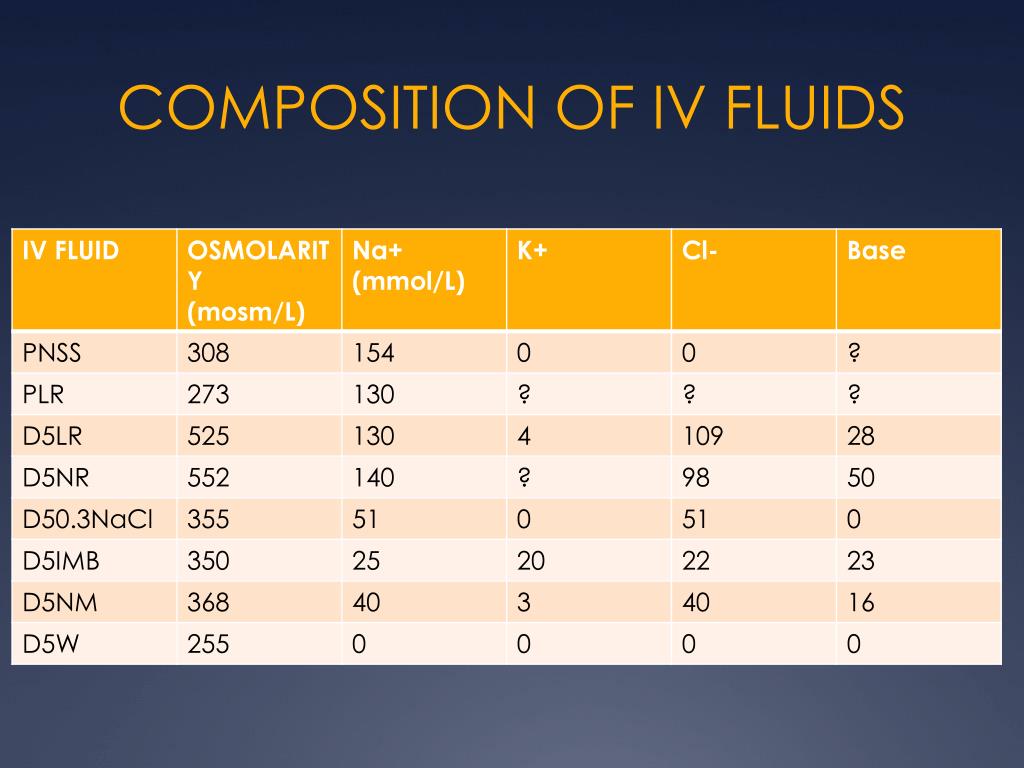
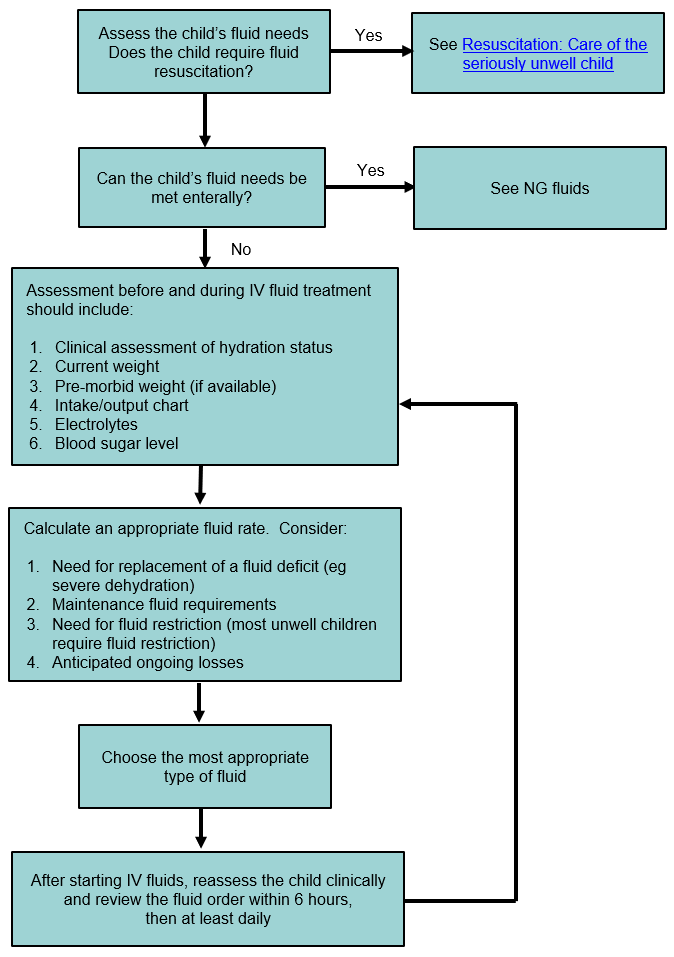
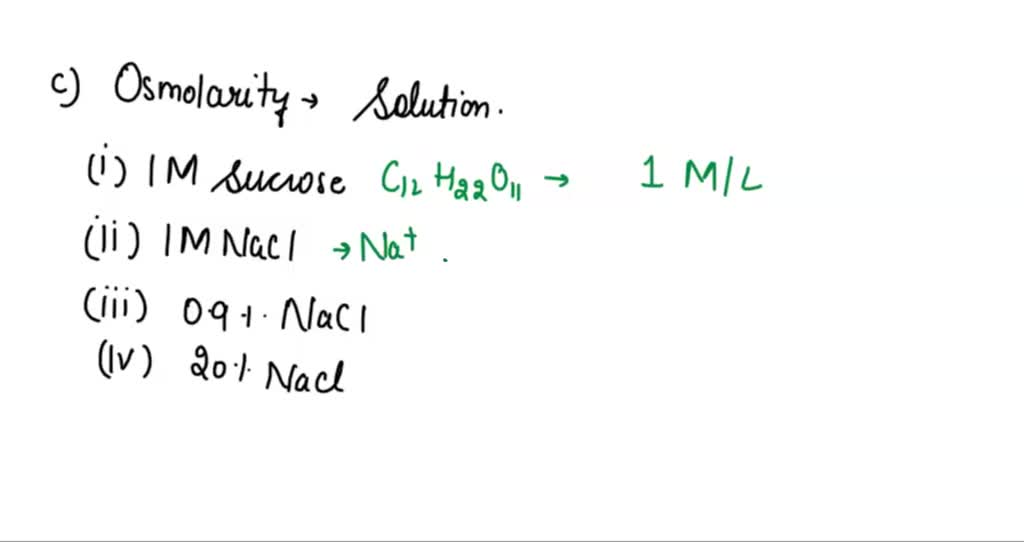What is the molarity (M) of a 5% dextrose (IV sugar solution) solution? (hint: molarity= number of moles of solute in 1 liter of 1000mL of solution). Show your calculations. | Homework.Study.com

Calculate your own osmolarity | Lab values and concentrations | Health & Medicine | Khan Academy - YouTube